Ticagrelor - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for ticagrelor and what is the scope of freedom to operate?
Ticagrelor
is the generic ingredient in two branded drugs marketed by Astrazeneca, Alembic, Alkem Labs Ltd, Amneal, Apotex, Changzhou Pharm, Dr Reddys, Hisun Pharm Hangzhou, Invagen Pharms, Macleods Pharms Ltd, MSN, Mylan, Prinston Inc, Sciegen Pharms Inc, Sunshine, Taro, and Watson Labs Inc, and is included in eighteen NDAs. There are three patents protecting this compound and two Paragraph IV challenges. Additional information is available in the individual branded drug profile pages.Ticagrelor has one hundred and forty-seven patent family members in forty-four countries.
There are twenty-one drug master file entries for ticagrelor. Twenty-five suppliers are listed for this compound. There is one tentative approval for this compound.
Summary for ticagrelor
| International Patents: | 147 |
| US Patents: | 3 |
| Tradenames: | 2 |
| Applicants: | 17 |
| NDAs: | 18 |
| Drug Master File Entries: | 21 |
| Finished Product Suppliers / Packagers: | 25 |
| Raw Ingredient (Bulk) Api Vendors: | 81 |
| Clinical Trials: | 461 |
| Patent Applications: | 3,245 |
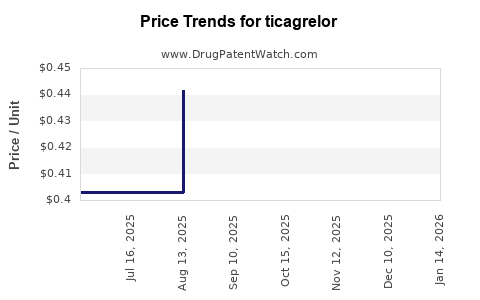
| Drug Prices: | Drug price trends for ticagrelor |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ticagrelor |
| What excipients (inactive ingredients) are in ticagrelor? | ticagrelor excipients list |
| DailyMed Link: | ticagrelor at DailyMed |
Recent Clinical Trials for ticagrelor
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Collegium Medicum w Bydgoszczy | PHASE4 |
| Ministry of Science and Higher Education, Poland | PHASE4 |
| University of Messina | PHASE4 |
Generic filers with tentative approvals for TICAGRELOR
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Get Started Free | ⤷ Get Started Free | 90MG | TABLET;ORAL |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Pharmacology for ticagrelor
| Drug Class | P2Y12 Platelet Inhibitor |
| Mechanism of Action | Cytochrome P450 3A4 Inhibitors P-Glycoprotein Inhibitors P2Y12 Receptor Antagonists |
| Physiological Effect | Decreased Platelet Aggregation |
Anatomical Therapeutic Chemical (ATC) Classes for ticagrelor
Paragraph IV (Patent) Challenges for TICAGRELOR
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| BRILINTA | Tablets | ticagrelor | 60 mg | 022433 | 3 | 2015-09-30 |
| BRILINTA | Tablets | ticagrelor | 90 mg | 022433 | 16 | 2015-07-20 |
US Patents and Regulatory Information for ticagrelor
Expired US Patents for ticagrelor
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astrazeneca | BRILINTA | ticagrelor | TABLET;ORAL | 022433-001 | Jul 20, 2011 | ⤷ Get Started Free | ⤷ Get Started Free |
| Astrazeneca | BRILINTA | ticagrelor | TABLET;ORAL | 022433-001 | Jul 20, 2011 | ⤷ Get Started Free | ⤷ Get Started Free |
| Astrazeneca | BRILINTA | ticagrelor | TABLET;ORAL | 022433-002 | Sep 3, 2015 | ⤷ Get Started Free | ⤷ Get Started Free |
| Astrazeneca | BRILINTA | ticagrelor | TABLET;ORAL | 022433-001 | Jul 20, 2011 | ⤷ Get Started Free | ⤷ Get Started Free |
| Astrazeneca | BRILINTA | ticagrelor | TABLET;ORAL | 022433-002 | Sep 3, 2015 | ⤷ Get Started Free | ⤷ Get Started Free |
| Astrazeneca | BRILINTA | ticagrelor | TABLET;ORAL | 022433-002 | Sep 3, 2015 | ⤷ Get Started Free | ⤷ Get Started Free |
| Astrazeneca | BRILINTA | ticagrelor | TABLET;ORAL | 022433-002 | Sep 3, 2015 | ⤷ Get Started Free | ⤷ Get Started Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for ticagrelor
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| AstraZeneca AB | Brilique | ticagrelor | EMEA/H/C/001241Brilique, co administered with acetylsalicylic acid (ASA), is indicated for the prevention of atherothrombotic events in adult patients withacute coronary syndromes (ACS) ora history of myocardial infarction (MI) and a high risk of developing an atherothrombotic eventBrilique, co-administered with acetyl salicylic acid (ASA), is indicated for the prevention of atherothrombotic events in adult patients with a history of myocardial infarction (MI occurred at least one year ago) and a high risk of developing an atherothrombotic event. | Authorised | no | no | no | 2010-12-03 | |
| AstraZeneca AB | Possia | ticagrelor | EMEA/H/C/002303Possia, co-administered with acetylsalicylic acid (ASA), is indicated for the prevention of atherothrombotic events in adult patients with acute coronary syndromes (unstable angina, non-ST-elevation myocardial infarction [NSTEMI] or ST-elevation myocardial infarction [STEMI]); including patients managed medically, and those who are managed with percutaneous coronary intervention (PCI) or coronary artery by-pass grafting (CABG). | Withdrawn | no | no | no | 2010-12-03 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for ticagrelor
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Russian Federation | 2012153069 | ⤷ Get Started Free | |
| Portugal | 2056832 | ⤷ Get Started Free | |
| Russian Federation | 2012121883 | ⤷ Get Started Free | |
| Indonesia | 29927 | ⤷ Get Started Free | |
| Hong Kong | 1061246 | ⤷ Get Started Free | |
| Spain | 2366902 | ⤷ Get Started Free | |
| Japan | 2020037564 | ⤷ Get Started Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ticagrelor
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1135391 | 2011/012 | Ireland | ⤷ Get Started Free | PRODUCT NAME: TICAGRELOR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTRATION NO/DATE: EU/1/10/655/001-006 20101203 |
| 1135391 | C01135391/01 | Switzerland | ⤷ Get Started Free | FORMER REPRESENTATIVE: BOHEST AG, CH |
| 1135391 | C300485 | Netherlands | ⤷ Get Started Free | PRODUCT NAME: TICAGRELOR OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/10/655/001-006 20101203 |
| 1135391 | SPC/GB11/016 | United Kingdom | ⤷ Get Started Free | PRODUCT NAME: TICAGRELOR OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF.; REGISTERED: UK EU/1/10/655/001 20101203; UK EU/1/10/655/002 20101203; UK EU/1/10/655/003 20101203; UK EU/1/10/655/004 20101203; UK EU/1/10/655/005 20101203; UK EU/1/10/655/006 20101203 |
| 1135391 | 11C0016 | France | ⤷ Get Started Free | PRODUCT NAME: TICAGRELOR OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELUI-CI; REGISTRATION NO/DATE IN FRANCE: EU/1/10/655/001 DU 20101203; REGISTRATION NO/DATE AT EEC: EU/1/10/655/001-006 DU 20101203 |
| 1135391 | PA2011004 | Lithuania | ⤷ Get Started Free | PRODUCT NAME: TICAGRELORUM; REGISTRATION NO/DATE: EU/1/10/655/001 2010 12 03 EU/1/10/655/002 2010 12 03 EU/1/10/655/003 2010 12 03 EU/1/10/655/004 2010 12 03 EU/1/10/655/005 2010 12 03 EU/1/10/655/00 20101203 |
| 1135391 | PA2011004,C1135391 | Lithuania | ⤷ Get Started Free | PRODUCT NAME: TICAGRELORUM; REGISTRATION NO/DATE: EU/1/10/655/001 2010 12 03 EU/1/10/655/002 2010 12 03 EU/1/10/655/003 2010 12 03 EU/1/10/655/004 2010 12 03 EU/1/10/655/005 2010 12 03 EU/1/10/655/00 20101203 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory of Ticagrelor
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.