Introduction to Alphagan P
Alphagan P, a brimonidine tartrate ophthalmic solution, is a crucial medication in the management of glaucoma and ocular hypertension. Developed by Allergan Inc., it has undergone significant formulation improvements to enhance efficacy and reduce side effects.
Mechanism of Action and Clinical Use
Alphagan P works as an alpha-adrenergic agonist, reducing intraocular pressure by decreasing the production of aqueous humor and increasing its drainage. This mechanism makes it effective for treating primary open-angle glaucoma and ocular hypertension[3].
Formulation Advancements
The newer formulation of Alphagan P, approved by the FDA in 2005, contains 0.1% brimonidine tartrate, a reduction from the original 0.2% concentration. This change was made to maintain efficacy while minimizing drug exposure and potential side effects. The new formulation also replaced the preservative benzalkonium chloride with Purite, a proprietary formulation of sodium perborate, which is less likely to cause ocular irritation[1][4].
Market Approval and Launch
Allergan received FDA approval for Alphagan P in 2005, with the goal of maximizing efficacy while reducing drug concentration. However, no specific market launch date was immediately set following the approval[1].
Financial Performance
Revenue Contribution
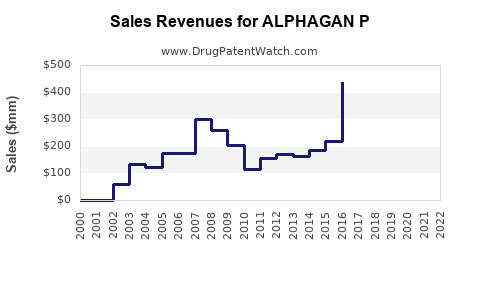
Alphagan P, along with other ophthalmic products like Combigan, contributed to Allergan's financial performance. In the fourth quarter of 2019, the net revenues from these products were $94.5 million, representing a 3.3% decrease from the prior year quarter. This decline is part of a broader financial context where Allergan reported full-year 2019 GAAP net revenues of $16.1 billion, a 1.9% increase from 2018[2].
Overall Financial Health of Allergan
In 2019, Allergan's financial health was marked by a 6.6% increase in fourth-quarter GAAP net revenues compared to the prior year, reaching $4.35 billion. However, the company also reported significant operating losses and expenses, including a 37.4% increase in selling, general, and administrative (SG&A) expenses and a 33.3% decrease in research and development (R&D) expenses[2].
Pricing and Affordability
The cost of Alphagan P can be significant, with an average retail price of $212.79 as of November 2024. However, patient assistance programs and advocacy groups like The Rx Advocates offer more affordable options, such as a fixed cost of $80 per month for Alphagan P alone or combined with other medications[3].
Market Dynamics and Competition
The ophthalmic market is competitive, with various products vying for market share. Alphagan P's unique formulation and reduced preservative content give it an edge over older formulations. However, other products like Lumigan (bimatoprost) also undergo formulation changes to improve efficacy and reduce side effects, indicating a dynamic market where innovation is key[4].
Patient Benefits and Adherence
The newer formulation of Alphagan P has been shown to reduce ocular allergic reactions and other adverse effects compared to its predecessor. This improvement enhances patient compliance and overall treatment outcomes. The use of Purite as a preservative further reduces the risk of ocular irritation, making the medication more tolerable for long-term use[4].
Secondary Uses and Versatility
Beyond its primary use in managing glaucoma and ocular hypertension, Alphagan P has shown efficacy in alleviating symptoms of dry eye syndrome. This versatility expands its therapeutic applications and makes it a valuable option for patients with multiple ocular health issues[3].
Adverse Effects and Interactions
While Alphagan P is generally well-tolerated, it can cause side effects such as ocular allergic reactions, although these are less frequent and severe compared to the older formulation. Healthcare providers must monitor patients for any adverse effects and adjust treatment plans accordingly[4].
Conclusion
Alphagan P is a significant player in the ophthalmic market, particularly for managing glaucoma and ocular hypertension. Its optimized formulation reduces drug exposure and side effects, making it a preferred choice for many patients. The financial performance of Alphagan P contributes to Allergan's overall revenue, although the market is competitive and dynamic. Patient assistance programs help make the medication more affordable, enhancing adherence and treatment outcomes.
Key Takeaways
- Optimized Formulation: Alphagan P's 0.1% brimonidine tartrate concentration reduces drug exposure without sacrificing efficacy.
- Preservative Change: The use of Purite instead of benzalkonium chloride reduces ocular irritation.
- Financial Contribution: Alphagan P contributes to Allergan's revenue, despite fluctuations in the market.
- Affordability: Patient assistance programs make Alphagan P more affordable for patients.
- Versatility: Alphagan P has secondary uses, including alleviating dry eye syndrome symptoms.
FAQs
1. What is Alphagan P used for?
Alphagan P is used to lower intraocular pressure in patients with primary open-angle glaucoma or ocular hypertension.
2. How does Alphagan P differ from its predecessor?
Alphagan P contains 0.1% brimonidine tartrate, a reduction from the original 0.2% concentration, and uses Purite instead of benzalkonium chloride as a preservative.
3. What are the potential side effects of Alphagan P?
Potential side effects include ocular allergic reactions, although these are less frequent and severe compared to the older formulation.
4. How much does Alphagan P cost?
The average retail cost of Alphagan P is $212.79, but patient assistance programs can reduce the cost to around $80 per month.
5. Is Alphagan P used for any other conditions?
Yes, Alphagan P has also shown efficacy in alleviating symptoms of dry eye syndrome.
Cited Sources:
- Ophthalmology Times: "New brimonidine formula approved for market"
- PR Newswire: "Allergan Reports Fourth Quarter and Full-Year 2019 Financial Results"
- The Rx Advocates: "Alphagan P Coupon & Assistance Programs"
- Review of Optometry: "Could Less Be More?"