ATRIPLA Drug Patent Profile
✉ Email this page to a colleague
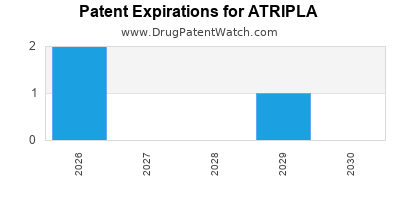
When do Atripla patents expire, and when can generic versions of Atripla launch?
Atripla is a drug marketed by Gilead Sciences and is included in one NDA. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has sixty-one patent family members in twenty-seven countries.
The generic ingredient in ATRIPLA is efavirenz; emtricitabine; tenofovir disoproxil fumarate. There are twenty-six drug master file entries for this compound. Seven suppliers are listed for this compound. Additional details are available on the efavirenz; emtricitabine; tenofovir disoproxil fumarate profile page.
DrugPatentWatch® Generic Entry Outlook for Atripla
Atripla was eligible for patent challenges on July 2, 2007.
There have been thirteen patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are two tentative approvals for the generic drug (efavirenz; emtricitabine; tenofovir disoproxil fumarate), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for ATRIPLA?
- What are the global sales for ATRIPLA?
- What is Average Wholesale Price for ATRIPLA?
Summary for ATRIPLA
| International Patents: | 61 |
| US Patents: | 3 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Clinical Trials: | 54 |
| Drug Prices: | Drug price information for ATRIPLA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for ATRIPLA |
| What excipients (inactive ingredients) are in ATRIPLA? | ATRIPLA excipients list |
| DailyMed Link: | ATRIPLA at DailyMed |

Recent Clinical Trials for ATRIPLA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Willem Daniel Francois Venter | Phase 1 |
| University of Cape Town | Phase 1 |
| Yu-Jay Corp. | Phase 3 |
Pharmacology for ATRIPLA
Paragraph IV (Patent) Challenges for ATRIPLA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ATRIPLA | Tablets | efavirenz; emtricitabine; tenofovir disoproxil fumarate | 600 mg/200 mg/300 mg | 021937 | 1 | 2008-12-29 |
US Patents and Regulatory Information for ATRIPLA
ATRIPLA is protected by five US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gilead Sciences | ATRIPLA | efavirenz; emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021937-001 | Jul 12, 2006 | DISCN | Yes | No | 9,018,192 | ⤷ Try for Free | ⤷ Try for Free | ||||
| Gilead Sciences | ATRIPLA | efavirenz; emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021937-001 | Jul 12, 2006 | DISCN | Yes | No | 9,545,414 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Gilead Sciences | ATRIPLA | efavirenz; emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021937-001 | Jul 12, 2006 | DISCN | Yes | No | 8,598,185 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ATRIPLA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Gilead Sciences | ATRIPLA | efavirenz; emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021937-001 | Jul 12, 2006 | 6,642,245*PED | ⤷ Try for Free |
| Gilead Sciences | ATRIPLA | efavirenz; emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021937-001 | Jul 12, 2006 | 8,716,264 | ⤷ Try for Free |
| Gilead Sciences | ATRIPLA | efavirenz; emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021937-001 | Jul 12, 2006 | 6,238,695 | ⤷ Try for Free |
| Gilead Sciences | ATRIPLA | efavirenz; emtricitabine; tenofovir disoproxil fumarate | TABLET;ORAL | 021937-001 | Jul 12, 2006 | 5,977,089*PED | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ATRIPLA
See the table below for patents covering ATRIPLA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Bulgaria | 62612 | ⤷ Try for Free | |
| Iceland | 7977 | Samsetningar og aðferð til samsettra meðferðar | ⤷ Try for Free |
| Norway | 325824 | ⤷ Try for Free | |
| Slovakia | 279542 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for ATRIPLA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0915894 | 05C0032 | France | ⤷ Try for Free | PRODUCT NAME: TENOFOVIR DISOPROXIL FUMARATE; EMTRICITABINE; REGISTRATION NO/DATE: EU/1/04/305/001 20050221 |
| 0915894 | 08C0020 | France | ⤷ Try for Free | PRODUCT NAME: EFAVIRENZ; EMTRICITABINE; TENOFOVIR DISOPROXIL FUMARATE; REGISTRATION NO/DATE: EU/1/07/430/001 20071213 |
| 0582455 | CA 2001 00014 | Denmark | ⤷ Try for Free | |
| 0915894 | CA 2005 00032 | Denmark | ⤷ Try for Free | |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for ATRIPLA
More… ↓


