CLINDAMYCIN PHOSPHATE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Clindamycin Phosphate, and when can generic versions of Clindamycin Phosphate launch?
Clindamycin Phosphate is a drug marketed by Glenmark Pharms Ltd, Padagis Israel, Taro, Fougera Pharms, Alembic, Amneal, Encube, Quagen, Solaris Pharma Corp, Zydus Lifesciences, Abraxis Pharm, Almaject, Bedford, Bristol Myers Squibb, Fresenius Kabi Usa, Hikma, Igi Labs Inc, Loch, Marsam Pharms Llc, Rising, Sagent Pharms Inc, Solopak, Teva Parenteral, Watson Labs, Boca Pharma Llc, Chartwell Rx, Encube Ethicals, Endo Operations, Fougera Pharms Inc, G And W Labs Inc, Novast Labs, Padagis Us, Pai Holdings Pharm, Taro Pharm Inds, Vintage Pharms, Xttrium Labs Inc, Epic Pharma Llc, Actavis Labs Ut Inc, Glenmark Speclt, Mylan Pharms Inc, Zydus Pharms, Actavis Mid Atlantic, Baxter Hlthcare Corp, Sandoz Inc, Abbott Labs, and Baxter Hlthcare. and is included in eighty-nine NDAs.
The generic ingredient in CLINDAMYCIN PHOSPHATE is clindamycin phosphate; tretinoin. There are fifty-five drug master file entries for this compound. Six suppliers are listed for this compound. Additional details are available on the clindamycin phosphate; tretinoin profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Clindamycin Phosphate
A generic version of CLINDAMYCIN PHOSPHATE was approved as clindamycin phosphate; tretinoin by ACTAVIS MID ATLANTIC on June 12th, 2015.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for CLINDAMYCIN PHOSPHATE?
- What are the global sales for CLINDAMYCIN PHOSPHATE?
- What is Average Wholesale Price for CLINDAMYCIN PHOSPHATE?
Summary for CLINDAMYCIN PHOSPHATE
| US Patents: | 0 |
| Applicants: | 46 |
| NDAs: | 89 |
| Finished Product Suppliers / Packagers: | 22 |
| Raw Ingredient (Bulk) Api Vendors: | 52 |
| Clinical Trials: | 30 |
| Patent Applications: | 2,414 |
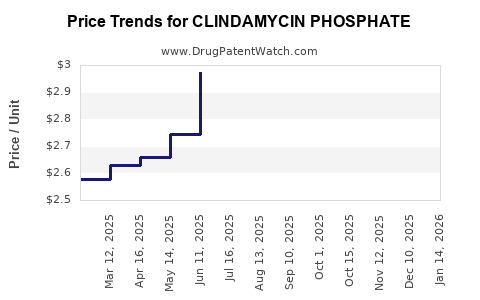
| Drug Prices: | Drug price information for CLINDAMYCIN PHOSPHATE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for CLINDAMYCIN PHOSPHATE |
| What excipients (inactive ingredients) are in CLINDAMYCIN PHOSPHATE? | CLINDAMYCIN PHOSPHATE excipients list |
| DailyMed Link: | CLINDAMYCIN PHOSPHATE at DailyMed |

Recent Clinical Trials for CLINDAMYCIN PHOSPHATE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Daré Bioscience, Inc. | Phase 1 |
| Memorial Sloan Kettering Cancer Center | Phase 2 |
| Daré Bioscience, Inc. | Phase 3 |
Pharmacology for CLINDAMYCIN PHOSPHATE
| Drug Class | Lincosamide Antibacterial |
| Physiological Effect | Decreased Sebaceous Gland Activity Neuromuscular Blockade |
Anatomical Therapeutic Chemical (ATC) Classes for CLINDAMYCIN PHOSPHATE
Paragraph IV (Patent) Challenges for CLINDAMYCIN PHOSPHATE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| CLINDESSE | Vaginal Cream | clindamycin phosphate | 2% | 050793 | 1 | 2015-02-05 |
US Patents and Regulatory Information for CLINDAMYCIN PHOSPHATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baxter Hlthcare | CLINDAMYCIN PHOSPHATE IN DEXTROSE 5% IN PLASTIC CONTAINER | clindamycin phosphate | INJECTABLE;INJECTION | 050648-001 | Dec 29, 1989 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Watson Labs | CLINDAMYCIN PHOSPHATE | clindamycin phosphate | INJECTABLE;INJECTION | 063079-001 | Mar 5, 1990 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Epic Pharma Llc | CLINDAMYCIN PHOSPHATE | clindamycin phosphate | SWAB;TOPICAL | 065513-001 | Jun 17, 2010 | AT | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Padagis Israel | CLINDAMYCIN PHOSPHATE AND BENZOYL PEROXIDE | benzoyl peroxide; clindamycin phosphate | GEL;TOPICAL | 090979-001 | Jun 26, 2012 | AB | RX | No | Yes | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| Endo Operations | CLINDAMYCIN PHOSPHATE | clindamycin phosphate | SOLUTION;TOPICAL | 203343-001 | May 29, 2015 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Rising | CLINDAMYCIN PHOSPHATE | clindamycin phosphate | INJECTABLE;INJECTION | 204748-001 | Oct 10, 2017 | DISCN | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | ||||
| Baxter Hlthcare Corp | CLINDAMYCIN PHOSPHATE IN 5% DEXTROSE IN PLASTIC CONTAINER | clindamycin phosphate | INJECTABLE;INJECTION | 208084-003 | Jun 28, 2017 | AP | RX | No | No | ⤷ Try for Free | ⤷ Try for Free | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Market Dynamics and Financial Trajectory for Clindamycin Phosphate
More… ↓


