LONSURF Drug Patent Profile
✉ Email this page to a colleague
When do Lonsurf patents expire, and when can generic versions of Lonsurf launch?
Lonsurf is a drug marketed by Taiho Oncology and is included in one NDA. There are six patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and eighteen patent family members in thirty-three countries.
The generic ingredient in LONSURF is tipiracil hydrochloride; trifluridine. There are two drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the tipiracil hydrochloride; trifluridine profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Lonsurf
A generic version of LONSURF was approved as tipiracil hydrochloride; trifluridine by NATCO on June 13th, 2023.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for LONSURF?
- What are the global sales for LONSURF?
- What is Average Wholesale Price for LONSURF?
Summary for LONSURF
| International Patents: | 118 |
| US Patents: | 6 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 13 |
| Clinical Trials: | 55 |
| Patent Applications: | 552 |
| Drug Prices: | Drug price information for LONSURF |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for LONSURF |
| What excipients (inactive ingredients) are in LONSURF? | LONSURF excipients list |
| DailyMed Link: | LONSURF at DailyMed |
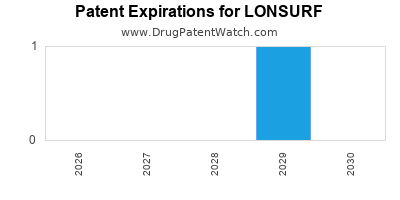

Paragraph IV (Patent) Challenges for LONSURF
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LONSURF | Tablets | tipiracil hydrochloride; trifluridine | 15 mg/6.14 mg and 20 mg/8.19 mg | 207981 | 4 | 2019-09-23 |
US Patents and Regulatory Information for LONSURF
LONSURF is protected by ten US patents and one FDA Regulatory Exclusivity.
Expired US Patents for LONSURF
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-001 | Sep 22, 2015 | 5,744,475 | ⤷ Try for Free |
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-002 | Sep 22, 2015 | 5,744,475 | ⤷ Try for Free |
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-001 | Sep 22, 2015 | 6,479,500 | ⤷ Try for Free |
| Taiho Oncology | LONSURF | tipiracil hydrochloride; trifluridine | TABLET;ORAL | 207981-002 | Sep 22, 2015 | 6,479,500 | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for LONSURF
When does loss-of-exclusivity occur for LONSURF?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 14282281
Patent: Stabilized crystal of tipiracil hydrochloride, and crystallization method for same
Estimated Expiration: ⤷ Try for Free
Patent: 17208215
Patent: Stable crystal form of tipiracil hydrochloride and crystallization method for the same
Estimated Expiration: ⤷ Try for Free
Patent: 18219967
Patent: Stable crystal form of tipiracil hydrochloride and crystallization method for the same
Estimated Expiration: ⤷ Try for Free
Brazil
Patent: 2015031619
Patent: forma cristalina estável de cloridrato de tipiracil e método de cristalização para a mesma
Estimated Expiration: ⤷ Try for Free
Canada
Patent: 14999
Patent: CRISTAL STABILISE DE CHLORHYDRATE DE TIPIRACIL, ET SON PROCEDE DE CRISTALLISATION (STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME)
Estimated Expiration: ⤷ Try for Free
Patent: 85006
Patent: CRISTAL STABILISE DE CHLORHYDRATE DE TIPIRACIL, ET SON PROCEDE DE CRISTALLISATION (STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME)
Estimated Expiration: ⤷ Try for Free
China
Patent: 4395307
Patent: STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME
Estimated Expiration: ⤷ Try for Free
Patent: 6967051
Patent: 地匹福林盐酸盐的稳定型晶体及其结晶化方法 (Stable crystal form of tipiracil hydrochloride and crystallization method for the same)
Estimated Expiration: ⤷ Try for Free
Patent: 9912573
Patent: 地匹福林盐酸盐的稳定型晶体及其结晶化方法 (STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME)
Estimated Expiration: ⤷ Try for Free
Patent: 0746403
Patent: 地匹福林盐酸盐的稳定型晶体及其结晶化方法 (STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME)
Estimated Expiration: ⤷ Try for Free
Croatia
Patent: 0211903
Estimated Expiration: ⤷ Try for Free
Cyprus
Patent: 24868
Estimated Expiration: ⤷ Try for Free
Denmark
Patent: 12255
Estimated Expiration: ⤷ Try for Free
European Patent Office
Patent: 12255
Patent: CRISTAL STABILISÉ DE CHLORHYDRATE DE TIPIRACIL, ET SON PROCÉDÉ DE CRISTALLISATION (STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME)
Estimated Expiration: ⤷ Try for Free
Patent: 05779
Patent: FORME CRISTALLINE STABLE DE CHLORHYDRATE DE TIPIRACIL ET SON PROCÉDÉ DE CRISTALLISATION (STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME)
Estimated Expiration: ⤷ Try for Free
Georgia, Republic of
Patent: 0186841
Patent: STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME
Estimated Expiration: ⤷ Try for Free
Patent: 0186875
Patent: STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLI- ZATION METHOD FOR SAME
Estimated Expiration: ⤷ Try for Free
Hong Kong
Patent: 03071
Patent: 地匹福林鹽酸鹽的穩定型晶體及其結晶化方法 (STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME)
Estimated Expiration: ⤷ Try for Free
Hungary
Patent: 57352
Estimated Expiration: ⤷ Try for Free
Japan
Patent: 2014203877
Patent: チピラシル塩酸塩の安定形結晶及びその結晶化方法
Estimated Expiration: ⤷ Try for Free
Patent: 64409
Estimated Expiration: ⤷ Try for Free
Patent: 26883
Estimated Expiration: ⤷ Try for Free
Patent: 18145204
Patent: チピラシル塩酸塩の安定形結晶及びその結晶化方法 (STABLE FORM CRYSTAL OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD THEREFOR)
Estimated Expiration: ⤷ Try for Free
Lithuania
Patent: 12255
Estimated Expiration: ⤷ Try for Free
Malaysia
Patent: 3410
Patent: STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME
Estimated Expiration: ⤷ Try for Free
Mexico
Patent: 15016986
Patent: FORMA DE CRISTAL ESTABLE DE CLORHIDRATO DE TIPIRACIL, Y METODO DE CRISTALIZACION DEL MISMO. (STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME.)
Estimated Expiration: ⤷ Try for Free
Morocco
Patent: 668
Patent: Cristal stabilisé de chlorhydrate de tipiracil, et son procédé de cristallisation
Estimated Expiration: ⤷ Try for Free
New Zealand
Patent: 4090
Patent: Stable crystal form of tipiracil hydrochloride and crystallization method for the same
Estimated Expiration: ⤷ Try for Free
Philippines
Patent: 015502809
Patent: STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME
Estimated Expiration: ⤷ Try for Free
Poland
Patent: 12255
Estimated Expiration: ⤷ Try for Free
Portugal
Patent: 12255
Estimated Expiration: ⤷ Try for Free
Russian Federation
Patent: 40417
Patent: СТАБИЛЬНАЯ КРИСТАЛЛИЧЕСКАЯ ФОРМА ТИПИРАЦИЛА ГИДРОХЛОРИДА И СПОСОБ ЕЕ КРИСТАЛЛИЗАЦИИ (STABLE CRYSTALLINE FORM OF TYPIRACYL HYDROCHLORIDE AND METHOD FOR ITS CRYSTALLIZATION)
Estimated Expiration: ⤷ Try for Free
Patent: 74441
Patent: СТАБИЛЬНАЯ КРИСТАЛЛИЧЕСКАЯ ФОРМА ТИПИРАЦИЛА ГИДРОХЛОРИДА И СПОСОБ ЕЕ КРИСТАЛЛИЗАЦИИ (STABLE CRYSTALLINE FORM OF TIPIRACIL HYDROCHLORIDE AND METHOD FOR ITS CRYSTALLIZATION)
Estimated Expiration: ⤷ Try for Free
Patent: 16100972
Patent: СТАБИЛЬНАЯ КРИСТАЛЛИЧЕСКАЯ ФОРМА ТИПИРАЦИЛА ГИДРОХЛОРИДА И СПОСОБ ЕЕ КРИСТАЛЛИЗАЦИИ
Estimated Expiration: ⤷ Try for Free
Serbia
Patent: 691
Patent: STABILNI KRISTALNI OBLIK TIPIRACIL HIDROHLORIDA I POSTUPAK KRISTALIZACIJE ISTOG (STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME)
Estimated Expiration: ⤷ Try for Free
Singapore
Patent: 201710916Y
Patent: STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME
Estimated Expiration: ⤷ Try for Free
Patent: 201912293U
Patent: STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME
Estimated Expiration: ⤷ Try for Free
Patent: 202007643S
Patent: STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME
Estimated Expiration: ⤷ Try for Free
Patent: 201509189S
Patent: STABLE CRYSTAL FORM OF TIPIRACIL HYDROCHLORIDE AND CRYSTALLIZATION METHOD FOR THE SAME
Estimated Expiration: ⤷ Try for Free
Slovenia
Patent: 12255
Estimated Expiration: ⤷ Try for Free
South Korea
Patent: 1806346
Estimated Expiration: ⤷ Try for Free
Patent: 160020560
Patent: 티피라실 염산염의 안정형 결정 및 그 결정화 방법 (STABILIZED CRYSTAL OF TIPIRACIL HYDROCHLORIDE, AND CRYSTALLIZATION METHOD FOR SAME)
Estimated Expiration: ⤷ Try for Free
Spain
Patent: 98408
Estimated Expiration: ⤷ Try for Free
Taiwan
Patent: 39592
Estimated Expiration: ⤷ Try for Free
Patent: 1534601
Patent: Stabilized crystal of tipiracil hydrochloride, and crystallization method for same
Estimated Expiration: ⤷ Try for Free
Ukraine
Patent: 2626
Patent: СТАБІЛЬНА КРИСТАЛІЧНА ФОРМА ТИПІРАЦИЛУ ГІДРОХЛОРИДУ І СПОСІБ ЇЇ КРИСТАЛІЗАЦІЇ
Estimated Expiration: ⤷ Try for Free
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering LONSURF around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Australia | 2017208215 | Stable crystal form of tipiracil hydrochloride and crystallization method for the same | ⤷ Try for Free |
| Australia | 772486 | ⤷ Try for Free | |
| Japan | 6882205 | ⤷ Try for Free | |
| Russian Federation | 2727598 | СПОСОБ ЛЕЧЕНИЯ БОЛЬНЫХ РАКОМ С ТЯЖЕЛОЙ ПОЧЕЧНОЙ НЕДОСТАТОЧНОСТЬЮ (METHOD OF TREATING PATIENTS WITH CANCER WITH SEVERE RENAL INSUFFICIENCY) | ⤷ Try for Free |
| Spain | 2203683 | ⤷ Try for Free | |
| Taiwan | 201536319 | Antitumor agent and antitumor effect enhancer | ⤷ Try for Free |
| World Intellectual Property Organization (WIPO) | 2014203877 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for LONSURF
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1849470 | 122017000052 | Germany | ⤷ Try for Free | PRODUCT NAME: TRIFLURIDIN IN KOMBINATION MIT TIPIRACIL ODER EINEM SALZ VON TIPIRACIL WIE TIPIRACIL-HYDROCHLORID; REGISTRATION NO/DATE: EU/1/16/1096 20160425 |
| 1849470 | 1790063-0 | Sweden | ⤷ Try for Free | PRODUCT NAME: TRIFLURIDINE IN COMBINATION WITH TIPIRACIL HYDROCHLORIDE; REG. NO/DATE: EU/1/16/1096 20160427 |
| 1849470 | 17C1028 | France | ⤷ Try for Free | PRODUCT NAME: TRIFLURIDINE/TIPIRACIL; REGISTRATION NO/DATE: EU/1/16/1096 20160427 |
| 1849470 | 2017/033 | Ireland | ⤷ Try for Free | PRODUCT NAME: TRIFLURIDINE IN COMBINATION WITH TIPIRACIL HYDROCHLORIDE; REGISTRATION NO/DATE: EU/1/16/1096 20160425 |
| 1849470 | 300889 | Netherlands | ⤷ Try for Free | PRODUCT NAME: TRIFLURIDINE IN COMBINATIE MET TIPIRACILHYDROCHLORIDE; REGISTRATION NO/DATE: EU/1/16/1096 20160427 |
| 1849470 | 34/2017 | Austria | ⤷ Try for Free | PRODUCT NAME: TRIFLURIDIN / TIPIRACIL ODER DEREN PHARMAZEUTISCH WIRKSAMEN SALZE; REGISTRATION NO/DATE: EU/1/16/1096 (MITTEILUNG) 20160427 |
| 1849470 | PA2017024,C1849470 | Lithuania | ⤷ Try for Free | PRODUCT NAME: TRIFLURIDINAS/TIPIRACILAS; REGISTRATION NO/DATE: EU/1/16/1096 20160425 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for LONSURF (Trifluridine/Tipiracil)
More… ↓


