POTASSIUM Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Potassium, and when can generic versions of Potassium launch?
Potassium is a drug marketed by Exela Pharma, Fresenius Kabi Usa, Hospira, Hexcel, Actavis Labs Fl Inc, Adare Pharms Inc, Amneal Pharms, Chartwell Molecular, Endo Operations, Glenmark Pharms Ltd, Granules, Lupin Ltd, Nesher Pharms, Novel Labs Inc, Padagis Us, Prinston Inc, Strides Pharma, Teva, Tris Pharma Inc, Zydus Pharms, Ajenat Pharms, Amneal, Epic Pharma Llc, Genus, Novitium Pharma, Rubicon, Abraxis Pharm, B Braun, Baxter Hlthcare, Gd Searle Llc, Intl Medication, Lilly, Luitpold, Miles, Nexus Pharms, Pharma Serve Ny, Watson Labs, Anda Repository, Apotex, Genus Lifesciences, Pharm Assoc, Sciegen Pharms Inc, Wes Pharma Inc, Ascent Pharms Inc, Aurobindo Pharma, Aurobindo Pharma Ltd, Breckenridge, Chartwell Rx, Copley Pharm, Guangzhou Novaken, Macleods Pharms Ltd, Mylan, Rising, Schering, Sigmapharm Labs Llc, Twi Pharms, Yichang Humanwell, Icu Medical Inc, Nexus, Ut Sw Medctr, Ani Pharms, Bionpharma, Hibrow Hlthcare, Teva Pharms Usa Inc, Torrent, Mission Pharmacal, Roxane, Am Regent, Cipla, Cmp Dev Llc, and Somerset Theraps Llc. and is included in one hundred and fifty-five NDAs. There is one patent protecting this drug.
The generic ingredient in POTASSIUM is potassium phosphate, dibasic; potassium phosphate, monobasic. There are two hundred and forty drug master file entries for this compound. Seven suppliers are listed for this compound. Additional details are available on the potassium phosphate, dibasic; potassium phosphate, monobasic profile page.
DrugPatentWatch® Generic Entry Outlook for Potassium
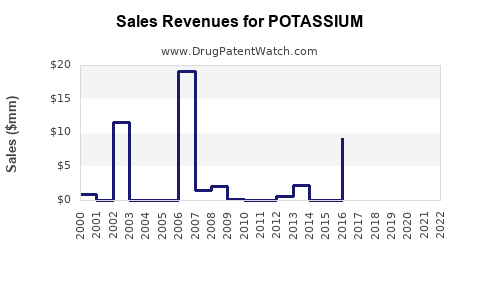
Annual sales in 2021 were $9mm indicating the motivation for generic entry (peak sales were $76mm in 2019).
Indicators of Generic Entry
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for POTASSIUM?
- What are the global sales for POTASSIUM?
- What is Average Wholesale Price for POTASSIUM?
Summary for POTASSIUM
| US Patents: | 0 |
| Applicants: | 71 |
| NDAs: | 155 |
| Drug Prices: | Drug price information for POTASSIUM |
| Drug Sales Revenues: | Drug sales revenues for POTASSIUM |
| DailyMed Link: | POTASSIUM at DailyMed |