Market Dynamics and Financial Trajectory for Pred Forte
Introduction
Pred Forte, a branded ophthalmic suspension used to treat steroid-responsive inflammation in the eye, has been a significant player in the pharmaceutical market. However, recent developments, including the launch of generic versions and changes in market dynamics, have significantly impacted its financial trajectory.
Market Overview
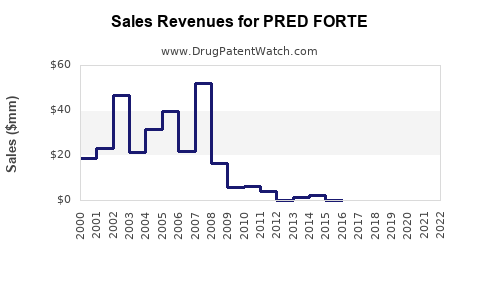
Pred Forte, developed by AbbVie Inc., has been widely prescribed for treating inflammation in various parts of the eye, including the palpebral and bulbar conjunctiva, cornea, and anterior segment of the globe. The drug has enjoyed substantial market share and revenue, with estimated annual sales of $198 million in the U.S. as of August 2024[3][4].
Generic Competition
A pivotal shift in the market dynamics for Pred Forte came with the launch of the first generic version by Lupin Ltd. in October 2024. This generic, Prednisolone Acetate Ophthalmic Suspension USP, 1%, is entitled to 180-day competitive generic therapy (CGT) exclusivity, which allows Lupin to be the sole generic provider during this period[3][4].
Impact on Pricing
The introduction of generic competition typically leads to a reduction in prices due to increased competition. However, the pricing dynamics for generic prednisolone acetate have been unusual. Historically, the price of generic prednisolone acetate was significantly lower than its branded counterpart. However, recent years have seen a convergence in prices, with the generic version's price approaching that of the branded Pred Forte. This is partly due to industry consolidation, reducing the number of manufacturers and thus competition, and regulatory challenges that have limited new entrants into the market[1].
Industry Consolidation
The pharmaceutical industry has experienced a wave of mergers and acquisitions, leading to fewer manufacturers producing fewer drugs. This consolidation has reduced competition, allowing companies to increase prices without fear of being undercut by competitors. For example, in the case of generic prednisolone acetate, there are currently only two producers in the U.S., Sandoz and Pacific Pharma, which has led to higher prices as these companies adjust their pricing strategies to maximize profits[1].
Regulatory and Supply Chain Factors
Regulatory challenges have also played a role in the pricing dynamics. The FDA's stringent approval process and the shutdown of noncompliant facilities have reduced the overall supply of generic medications, including prednisolone acetate. This reduction in supply, coupled with increased demand due to the Affordable Care Act (ACA) bringing more consumers into the prescription drug market, has driven up prices[1].
Financial Trajectory
Branded Pred Forte
The financial trajectory of Pred Forte has been impacted by the rising costs and the impending generic competition. Despite the high demand, the introduction of a generic version is expected to erode the market share and revenue of the branded product. In the past, Allergan (now part of AbbVie) increased the price of Pred Forte by 80% in response to the rising prices of the generic equivalent, highlighting the competitive pricing strategies in the market[1].
Generic Prednisolone Acetate
Lupin's launch of the generic version is expected to be a significant revenue generator, given the 180-day exclusivity period. With annual sales of $198 million for Pred Forte, Lupin is poised to capture a substantial share of this market. The company's stock has already seen a surge following the announcement, reflecting investor confidence in the financial potential of this new product[3][4].
Market Exclusivity and Competitive Advantage
The 180-day CGT exclusivity granted to Lupin provides a competitive advantage, allowing the company to establish itself as the primary generic provider before other generics enter the market. This exclusivity period is crucial for Lupin to maximize its market share and revenue before the market becomes more competitive[3][4].
Consumer and Healthcare Impact
The launch of the generic version of Pred Forte is expected to expand access to affordable healthcare for patients requiring treatment for steroid-responsive inflammation. Despite the initial high prices of the generic, the long-term effect is likely to be a reduction in costs for patients and healthcare systems as more generic competitors enter the market[3][4].
Conclusion
The market dynamics and financial trajectory for Pred Forte are undergoing significant changes with the introduction of generic competition. While the branded product faces challenges from rising costs and generic competition, the launch of the generic version by Lupin is expected to bring in substantial revenue and expand access to affordable healthcare.
Key Takeaways
- Generic Competition: The launch of the first generic version of Pred Forte by Lupin Ltd. marks a significant shift in the market.
- Pricing Dynamics: The prices of generic prednisolone acetate have converged with those of the branded product due to industry consolidation and regulatory factors.
- Regulatory and Supply Chain: FDA regulations and supply chain issues have reduced the overall supply, driving up prices.
- Financial Trajectory: The branded product faces revenue erosion, while the generic version is expected to generate substantial revenue.
- Market Exclusivity: Lupin's 180-day CGT exclusivity provides a competitive advantage in the market.
FAQs
Q: What is Pred Forte used for?
A: Pred Forte is used to treat steroid-responsive inflammation in the eye, including the palpebral and bulbar conjunctiva, cornea, and anterior segment of the globe.
Q: Who launched the first generic version of Pred Forte?
A: Lupin Ltd. launched the first generic version of Pred Forte in October 2024.
Q: What is the significance of the 180-day CGT exclusivity for Lupin?
A: The 180-day CGT exclusivity allows Lupin to be the sole generic provider of Pred Forte, giving the company a competitive advantage and the opportunity to maximize its market share and revenue.
Q: How have industry consolidation and regulatory factors affected the pricing of generic prednisolone acetate?
A: Industry consolidation has reduced competition, and regulatory challenges have limited new entrants, leading to higher prices for the generic version.
Q: What is the estimated annual sales revenue for Pred Forte in the U.S.?
A: The estimated annual sales revenue for Pred Forte in the U.S. is $198 million as of August 2024.
Sources
- CRSToday: "Bargain No More"
- Europarl.europa.eu: "Doslovný záznam ze zasedání - Středa, 27. listopadu 2024"
- CNBCTV18: "Lupin gains on launch of first generic of Pred Forte ophthalmic suspension in the US"
- Lupin.com: "Lupin Launches First Generic of Pred Forte® in the United States"