SAXENDA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Saxenda, and what generic alternatives are available?
Saxenda is a drug marketed by Novo and is included in one NDA. There are eighteen patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and sixty-six patent family members in twenty-nine countries.
The generic ingredient in SAXENDA is liraglutide. There are seven drug master file entries for this compound. Five suppliers are listed for this compound. Additional details are available on the liraglutide profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Saxenda
A generic version of SAXENDA was approved as liraglutide by HIKMA on December 23rd, 2024.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for SAXENDA?
- What are the global sales for SAXENDA?
- What is Average Wholesale Price for SAXENDA?
Summary for SAXENDA
| International Patents: | 166 |
| US Patents: | 18 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 2 |
| Raw Ingredient (Bulk) Api Vendors: | 27 |
| Clinical Trials: | 33 |
| Patent Applications: | 2,996 |
| Drug Prices: | Drug price information for SAXENDA |
| Drug Sales Revenues: | Drug sales revenues for SAXENDA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for SAXENDA |
| What excipients (inactive ingredients) are in SAXENDA? | SAXENDA excipients list |
| DailyMed Link: | SAXENDA at DailyMed |
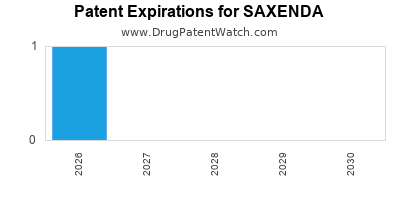
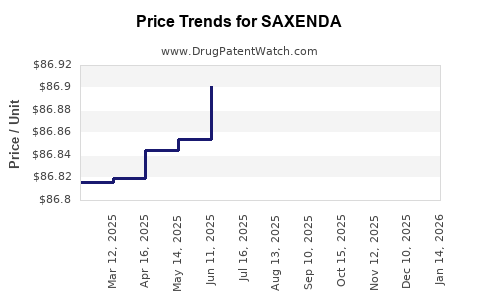
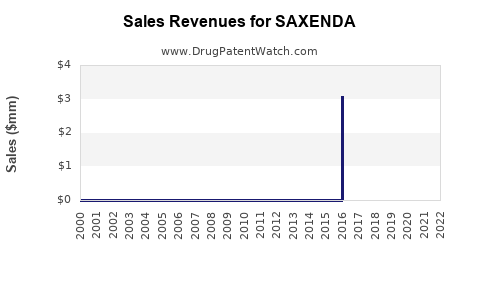
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for SAXENDA
Generic Entry Date for SAXENDA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
SOLUTION;SUBCUTANEOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for SAXENDA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Chicago | Early Phase 1 |
| Sciwind Biosciences APAC CO Pty. Ltd. | Phase 2 |
| Hangzhou Sciwind Biosciences Co., Ltd. | Phase 2 |
Pharmacology for SAXENDA
| Drug Class | GLP-1 Receptor Agonist |
| Mechanism of Action | Glucagon-like Peptide-1 (GLP-1) Agonists |
Paragraph IV (Patent) Challenges for SAXENDA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| SAXENDA | Injection | liraglutide | 18 mg/3 mL prefilled syringe | 206321 | 1 | 2021-08-16 |
US Patents and Regulatory Information for SAXENDA
SAXENDA is protected by eighteen US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of SAXENDA is ⤷ Try for Free.
This potential generic entry date is based on patent ⤷ Try for Free.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Novo | SAXENDA | liraglutide | SOLUTION;SUBCUTANEOUS | 206321-001 | Dec 23, 2014 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Novo | SAXENDA | liraglutide | SOLUTION;SUBCUTANEOUS | 206321-001 | Dec 23, 2014 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Novo | SAXENDA | liraglutide | SOLUTION;SUBCUTANEOUS | 206321-001 | Dec 23, 2014 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Novo | SAXENDA | liraglutide | SOLUTION;SUBCUTANEOUS | 206321-001 | Dec 23, 2014 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Novo | SAXENDA | liraglutide | SOLUTION;SUBCUTANEOUS | 206321-001 | Dec 23, 2014 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Novo | SAXENDA | liraglutide | SOLUTION;SUBCUTANEOUS | 206321-001 | Dec 23, 2014 | RX | Yes | Yes | ⤷ Try for Free | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for SAXENDA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Novo | SAXENDA | liraglutide | SOLUTION;SUBCUTANEOUS | 206321-001 | Dec 23, 2014 | ⤷ Try for Free | ⤷ Try for Free |
| Novo | SAXENDA | liraglutide | SOLUTION;SUBCUTANEOUS | 206321-001 | Dec 23, 2014 | ⤷ Try for Free | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for SAXENDA
See the table below for patents covering SAXENDA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Chile | 2018002515 | Liraglutida en afecciones cardiovasculares. | ⤷ Try for Free |
| Japan | 3149958 | ⤷ Try for Free | |
| Norway | 2009027 | ⤷ Try for Free | |
| Russian Federation | 2005102605 | СИСТЕМА ДЛЯ УСТАНОВКИ ИГЛЫ И СПОСОБ КРЕПЛЕНИЯ УЗЛА С ИГЛОЙ | ⤷ Try for Free |
| South Korea | 100556067 | ⤷ Try for Free | |
| Spain | 2334808 | ⤷ Try for Free | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for SAXENDA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0944648 | 09C0054 | France | ⤷ Try for Free | PRODUCT NAME: LIRAGLUTIDE; REGISTRATION NO/DATE IN FRANCE: EU/1/09/529/001 DU 20090630; REGISTRATION NO/DATE AT EEC: EU/1/09/529/001 DU 20090630 |
| 2209800 | 122014000114 | Germany | ⤷ Try for Free | PRODUCT NAME: KOMBINATION VON INSULIN DEGLUDEC UND LIRAGLUTIDE; REGISTRATION NO/DATE: EU/1/14/947 20140918 |
| 2209800 | 14C0085 | France | ⤷ Try for Free | PRODUCT NAME: LIRAGLUTIDE ET INSULINE DEGLUDEC; REGISTRATION NO/DATE: EU/1/14/947 20140918 |
| 2597103 | 2017/015 | Ireland | ⤷ Try for Free | PRODUCT NAME: COMBINATION OF INSULIN DEGLUDEC AND LIRAGLUTIDE; NAT REGISTRATION NO/DATE: EU/1/14/947 20140918; FIRST REGISTRATION NO/DATE: 65041 20140912 |
| 0944648 | C00944648/01 | Switzerland | ⤷ Try for Free | PRODUCT NAME: LIRAGLUTID; REGISTRATION NO/DATE: SWISSMEDIC 59329 11.12.2009 |
| 0944648 | SPC034/2009 | Ireland | ⤷ Try for Free | SPC034/2009: 20101001, EXPIRES: 20220821 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for SAXENDA (Liraglutide)
More… ↓


