Introduction to Dexamethasone
Dexamethasone is a potent anti-inflammatory synthetic adrenal corticosteroid used to treat a wide range of inflammatory and immune-related conditions. Its applications include allergic disorders, skin diseases, adrenal problems, arthritis, severe allergies, asthma, blood or bone marrow problems, and more recently, as a lifesaving treatment for critically ill COVID-19 patients[1][4][5].
Market Size and Forecast
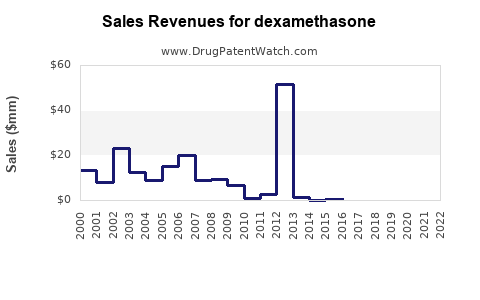
The global dexamethasone market is expected to experience significant growth over the forecast period of 2021 to 2028. According to market analyses, the dexamethasone market is projected to reach USD 4,634.01 million by 2028, growing at a Compound Annual Growth Rate (CAGR) of 6.65% during this period[1][4].
Key Drivers of Market Growth
Several factors are driving the growth of the dexamethasone market:
COVID-19 Impact
The rapid adoption of dexamethasone as a lifesaving therapy for severely ill COVID-19 patients has been a major driver. Studies, such as the RECOVERY trial, have shown that dexamethasone reduces 28-day mortality among patients receiving invasive mechanical ventilation or oxygen[5].
Increasing Prevalence of Inflammatory Conditions
The rising incidence of various inflammatory ailments, including cancer, allergic disorders, skin conditions, ulcerative colitis, arthritis, lupus, and psoriasis, is fueling the market growth[4].
Robust Investment in Research and Development
Significant investments in research and development are contributing to the market's expansion. This includes the development of new formulations and applications, such as the recent FDA approval for ciprofloxacin and dexamethasone otic suspension[1][3].
Public Awareness and Pandemic Impact
High incidences of viral disease outbreaks and increasing public awareness regarding the use of dexamethasone are also enhancing market growth[1][4].
Market Segmentation
The dexamethasone market is segmented based on several criteria:
Formulation Type
Dexamethasone is available in various forms, including oral tablets, oral solutions, eye drops, ear drops, injectable solutions, and intraocular solutions[2].
Patient Type
The market is segmented based on patient type, including adults and children, each with different dosage requirements and treatment plans[2].
Application
The primary applications include anti-inflammatory and immunosuppressant effects, respiratory distress syndrome, COVID-19 treatment, and others. The COVID-19 sector is expected to be the fastest-growing segment due to its proven benefits in reducing mortality among critically ill patients[4].
End Users
The market is bifurcated into hospital pharmacies, clinics, pharmacy and drug stores, and online pharmacies. Hospitals are expected to hold the majority of the market share due to increased hospital admissions for various diseases, including COVID-19[4].
Geographical Analysis
North America
North America leads the dexamethasone market due to the rapid increase in COVID-19 cases and the high prevalence of diseases in the region[1][4].
Asia-Pacific
The Asia-Pacific region is expected to expand at a significant growth rate due to the rising prevalence of viral disease outbreaks and the strong presence of major dexamethasone manufacturers in this region[1][4].
Financial Trajectory
Cost Factors
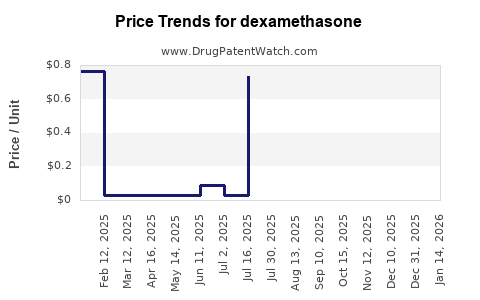
The cost of dexamethasone can vary based on several factors, including treatment plans, insurance coverage, pharmacy choices, and the need for additional equipment like needles and syringes. Generic versions of dexamethasone are generally more affordable than brand-name alternatives like DexPak and Decadron[2].
Financial Assistance and Savings
Patients can lower the cost of dexamethasone through various means, such as getting a 90-day supply, using mail-order pharmacies, and utilizing savings programs and coupons. Organizations and websites provide resources for drug assistance programs and savings cards[2].
Revenue Streams
The approval of new dexamethasone formulations, such as the ciprofloxacin and dexamethasone otic suspension, can lead to increased revenue streams for pharmaceutical companies. While generics typically offer lower margins than branded drugs, they can achieve significant volume sales[3].
Challenges and Restraints
Side Effects
The various side effects associated with dexamethasone, such as depression, euphoria, and hypertension, are expected to impede market growth. These adverse effects can deter some patients and healthcare providers from using the drug[1][4].
Preventive Measures
The development of vaccines and other preventive measures could challenge the growth of the dexamethasone market by reducing the need for corticosteroid treatments[1].
Key Takeaways
- The global dexamethasone market is projected to grow significantly due to its use in treating COVID-19 and other inflammatory conditions.
- North America and Asia-Pacific are key regions driving market growth.
- The market is segmented based on formulation type, patient type, application, and end users.
- Financial assistance programs and generic versions can help reduce the cost of dexamethasone.
- Side effects and preventive measures are potential restraints on market growth.
FAQs
-
What are the primary applications of dexamethasone?
- Dexamethasone is used for anti-inflammatory and immunosuppressant effects, respiratory distress syndrome, COVID-19 treatment, and other inflammatory conditions[4].
-
How does the cost of dexamethasone vary?
- The cost of dexamethasone can vary based on treatment plans, insurance coverage, pharmacy choices, and the need for additional equipment[2].
-
Is dexamethasone covered by Medicare?
- Dexamethasone may be covered by Medicare, but coverage and costs depend on the specific Medicare plan[2].
-
What are the potential side effects of dexamethasone?
- Potential side effects include depression, euphoria, hypertension, and other adverse effects[1][4].
-
How has COVID-19 impacted the dexamethasone market?
- The COVID-19 pandemic has significantly increased the demand for dexamethasone due to its effectiveness in reducing mortality among critically ill patients[1][4][5].
Cited Sources
- Data Bridge Market Research: Global Dexamethasone Market – Industry Trends and Forecast to 2028.
- Medical News Today: Dexamethasone cost: Financial assistance, savings, more.
- Stock Titan: Amneal Receives U.S. FDA Approval for Ciprofloxacin and Dexamethasone Otic Suspension.
- Verified Market Research: Dexamethasone Market Size, Share, Trends, Opportunities & Forecast.
- MedRxiv: Effect of Dexamethasone in Hospitalized Patients with COVID-19.