MOXEZA Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Moxeza, and what generic alternatives are available?
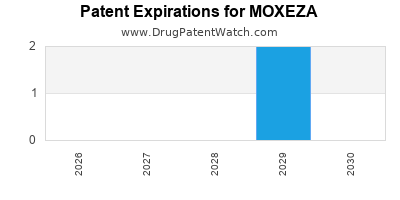
Moxeza is a drug marketed by Harrow Eye and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has twenty-three patent family members in fifteen countries.
The generic ingredient in MOXEZA is moxifloxacin hydrochloride. There are eighteen drug master file entries for this compound. Thirty-four suppliers are listed for this compound. Additional details are available on the moxifloxacin hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Moxeza
A generic version of MOXEZA was approved as moxifloxacin hydrochloride by TEVA PHARMS USA on February 18th, 2014.
AI Research Assistant
Questions you can ask:
- What is the 5 year forecast for MOXEZA?
- What are the global sales for MOXEZA?
- What is Average Wholesale Price for MOXEZA?
Summary for MOXEZA
| International Patents: | 23 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 111 |
| Clinical Trials: | 1 |
| Patent Applications: | 1,698 |
| Drug Prices: | Drug price information for MOXEZA |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for MOXEZA |
| What excipients (inactive ingredients) are in MOXEZA? | MOXEZA excipients list |
| DailyMed Link: | MOXEZA at DailyMed |

Recent Clinical Trials for MOXEZA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Usona Institute | Phase 1 |
Paragraph IV (Patent) Challenges for MOXEZA
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| MOXEZA | Ophthalmic Solution | moxifloxacin hydrochloride | 0.5% | 022428 | 1 | 2012-02-29 |
US Patents and Regulatory Information for MOXEZA
MOXEZA is protected by two US patents.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Harrow Eye | MOXEZA | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 022428-001 | Nov 19, 2010 | DISCN | Yes | No | 9,114,168 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| Harrow Eye | MOXEZA | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 022428-001 | Nov 19, 2010 | DISCN | Yes | No | 8,450,311 | ⤷ Try for Free | Y | ⤷ Try for Free | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for MOXEZA
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Harrow Eye | MOXEZA | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 022428-001 | Nov 19, 2010 | 4,990,517*PED | ⤷ Try for Free |
| Harrow Eye | MOXEZA | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 022428-001 | Nov 19, 2010 | 5,607,942*PED | ⤷ Try for Free |
| Harrow Eye | MOXEZA | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 022428-001 | Nov 19, 2010 | 7,671,070*PED | ⤷ Try for Free |
| Harrow Eye | MOXEZA | moxifloxacin hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 022428-001 | Nov 19, 2010 | 6,716,830*PED | ⤷ Try for Free |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for MOXEZA
See the table below for patents covering MOXEZA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2774207 | ⤷ Try for Free | |
| European Patent Office | 1117406 | ⤷ Try for Free | |
| Hong Kong | 1150756 | ⤷ Try for Free | |
| Mexico | 2010012933 | COMPOSICIONES FARMACEUTICAS QUE CONTIENEN UNA DROGA ANTIBIOTICA DE FLUOROQUINOLONA. (PHARMACEUTICAL COMPOSITIONS CONTAINING A FLUOROQUINOLONE ANTIBIOTIC DRUG.) | ⤷ Try for Free |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for MOXEZA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0350733 | 2001C/030 | Belgium | ⤷ Try for Free | PRODUCT NAME: MOXIFLOXACINE CHLORHYDRATE (CORRESPONDANT A MOXIFLOXACINE); NATL. REGISTRATION NO/DATE: 187 IS 328 F 3 20010507; FIRST REGISTRATION: DE 45263.00.00 19990621 |
| 0350733 | C300111 | Netherlands | ⤷ Try for Free | PRODUCT NAME: MOXIFLOXACINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER MOXIFLOXACINEHYDROCHLORIDE; NAT. REGISTRATION NO/DATE: RVG 28118 RVG 28119 20021017; FIRST REGISTRATION: DE 45263.00.00 19990621 |
| 0780390 | PA2004012,C0780390 | Lithuania | ⤷ Try for Free | PRODUCT NAME: MOXIFLOXACINI HYDROCHLORIDUM (1-CIKLOPROPIL-6-FLUOR-1,4-DIHIDRO-8-METOKSI-7-((4AS, 7AS)-OKTAHIDRO-6H-PIROLO(3,4-B)PIRIDIN-6-IL)-4-OKSO-3-CHINOLINKARBOKSIRUGSTIES HIDROCHLORIDAS); REGISTRATION NO/DATE: 04/8383/3 20040309 |
| 0350733 | SPC/GB03/034 | United Kingdom | ⤷ Try for Free | PRODUCT NAME: MOXIFLOXACIN AND PHARMACEUTICALLY USABLE HYDRATES AND ACID ADDITION SALTS THEREOF AND THE ALKALI METAL, ALKALINE EARTH METAL, SILVER AND GUANIDINIUM SALTS OF THE UNDERLYING CARBOXYLIC ACIDS AND THE RACEMATES THEREOF; REGISTERED: DE 45263.00.00 19990621; UK PL 000 10/0291 20030313 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
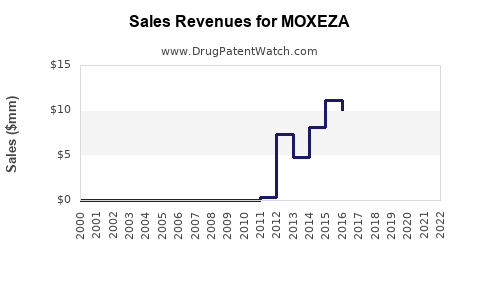
Market Dynamics and Financial Trajectory for Moxeza
More… ↓


