Introduction
Potassium chloride, a crucial compound in various industries, including pharmaceuticals, agriculture, and industrial applications, has seen significant market dynamics over the years. This article delves into the current market situation, growth drivers, challenges, and the financial trajectory of potassium chloride, particularly focusing on its pharmaceutical grade.
Global Market Overview
The global potassium chloride market is diverse and expansive, with applications spanning multiple sectors.
Market Size and Growth
As of 2024, the global Pharma Grade Potassium Chloride market is estimated to be worth US$ 15 million and is projected to grow to US$ 22 million by 2032, with a Compound Annual Growth Rate (CAGR) of 6.5% during the review period[1].
For the broader potassium chloride market, it is expected to reach US$ 133.50 billion by 2030, growing at a CAGR of 10.50% from 2024 to 2030[3].
Regional Outlook
Europe is the largest production area for pharma-grade potassium chloride, accounting for about 30% of the global market, followed by China and the rest of Asia, which together hold over 50% of the market share[1].
In the United States, the potassium chloride market holds a significant share, particularly in the North America industry, with the U.S. market accounting for 86.1% of the region's industry. China is also a major player, with its market expected to grow at a CAGR of 8.9% from 2020 to 2027[4].
Applications of Potassium Chloride
Pharmaceutical Sector
In the pharmaceutical sector, potassium chloride is used in various forms, including injectable solutions and hemodialysis solutions. It is a critical component in the formulation of certain medications and medical treatments. The pharmaceutical application segment was the leading segment in 2023, accounting for a significant market share[1].
Agricultural Sector
Potassium chloride is widely used as a fertilizer to improve soil fertility and enhance crop yields. The increasing demand for food due to a growing global population is driving the use of potassium chloride in agriculture. This sector is projected to record a 6.1% CAGR from 2020 to 2027[2].
Industrial Sector
The industrial sector, including applications in oil & gas, water treatment, and food processing, also contributes significantly to the demand for potassium chloride. The trend of using potassium chloride as feedstock raw materials is an additional factor driving market growth[3].
Market Drivers
Increasing Demand in Fertilizers
The increasing adoption of potassium chloride in fertilizers is a key factor driving the growth of the global market. This is due to its essential role in improving soil fertility and enhancing crop yields[3].
Pharmaceutical and Medical Applications
The demand for potassium chloride in pharmaceuticals, particularly for injectable solutions and hemodialysis, continues to drive market growth. For instance, high-purity pharmaceutical salts played a crucial role in the administration of coronavirus vaccines during the pandemic[5].
Industrial Expansion
Industrial expansion, especially in countries like China, is boosting the demand for potassium chloride in sectors such as water treatment, food processing, and oil & gas[4].
Market Constraints and Challenges
Economic Impact of COVID-19
The COVID-19 pandemic had a significant impact on the global potassium chloride market, leading to a readjustment in market size and growth projections. Despite this, the market is expected to recover and grow steadily in the post-pandemic period[1].
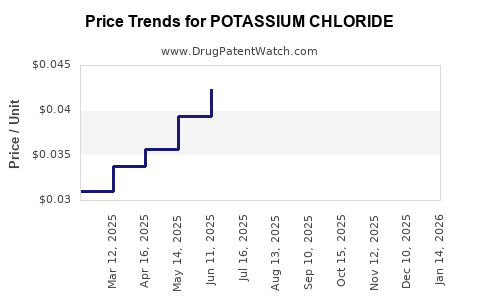
Price Volatility
One of the key factors hindering the growth of the global potassium chloride market is the shifting prices of potassium chloride. This volatility can affect production costs and profitability for manufacturers[3].
Competition
The market is fairly consolidated but is expected to see fragmentation as emerging players try to capitalize on opportunities. This competition, especially in regions like China, can be fierce and challenging for market participants[4].
Key Players
Major players in the potassium chloride market include ICL (ICL-IP), K+S, and Morton. These companies have a significant presence in the global market and are involved in various strategic initiatives to maintain their market share and competitiveness[1].
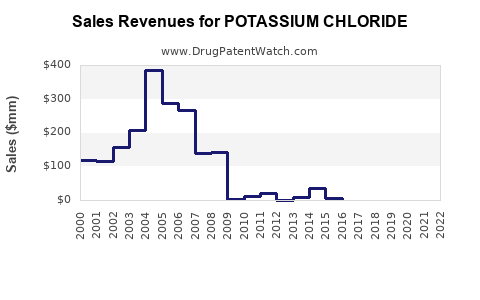
Financial Performance and Outlook
Revenue Projections
The global potassium chloride market is projected to reach significant revenue milestones. For instance, the broader market is expected to reach US$ 133.50 billion by 2030, while the pharma-grade segment is forecasted to reach US$ 22 million by 2032[1][3].
Cost Optimization
Companies like K+S are focusing on cost optimization and efficiency improvements. For example, K+S generated more than €150 million from synergies in procurement, logistics, production, and sales and marketing, and reduced the average cost of potash production to less than €200 per tonne[5].
Strategic Initiatives
Key players are engaging in strategic initiatives such as production facility expansions, acquisitions, and joint ventures to enhance their market position. K+S, for instance, sold its Americas operating unit and incorporated its waste management business into a joint venture to streamline operations and reduce costs[5].
Regional Market Dynamics
United States
The United States is a prominent producer and consumer of potassium chloride, with the market driven by its use in agriculture, pharmaceuticals, water treatment, and food processing. The U.S. market holds an 86.1% share in the North America industry[4].
China
China is expected to be the dominant market for potassium chloride in Asia from 2023 to 2033. The country's focus on industrial expansion, particularly in sectors like water treatment, food processing, and agriculture, is driving the demand for potassium chloride[4].
Europe
Europe, particularly Germany, is forecast to grow at a CAGR of approximately 3.8% from 2020 to 2027. The region is a significant production hub for pharma-grade potassium chloride[2].
Conclusion
The potassium chloride market, especially the pharma-grade segment, is poised for steady growth driven by increasing demand in fertilizers, pharmaceutical applications, and industrial expansion. Despite challenges such as price volatility and competition, key players are implementing strategic initiatives to maintain market share and drive growth.
Key Takeaways
- The global pharma-grade potassium chloride market is expected to grow at a CAGR of 6.5% from 2024 to 2032.
- The broader potassium chloride market is projected to reach US$ 133.50 billion by 2030.
- Key applications include pharmaceuticals, agriculture, and industrial sectors.
- Major players are focusing on cost optimization and strategic initiatives to enhance market position.
- Regional markets such as the United States, China, and Europe are significant contributors to the global market.
FAQs
Q: What is the projected market size of the global pharma-grade potassium chloride market by 2032?
A: The global pharma-grade potassium chloride market is forecasted to reach US$ 22 million by 2032[1].
Q: Which region is the largest production area for pharma-grade potassium chloride?
A: Europe is the largest production area for pharma-grade potassium chloride, accounting for about 30% of the global market[1].
Q: What are the primary drivers of the global potassium chloride market?
A: The primary drivers include increasing adoption in fertilizers, demand from pharmaceutical and medical applications, and industrial expansion[3].
Q: How has the COVID-19 pandemic affected the potassium chloride market?
A: The pandemic led to a readjustment in market size and growth projections, but the market is expected to recover and grow steadily in the post-pandemic period[1].
Q: Who are the major players in the potassium chloride market?
A: Major players include ICL (ICL-IP), K+S, and Morton[1].
Sources
- Pharma Grade Potassium Chloride Market Dynamics till 2032 | Growth Drivers, Market Constraints and Challenges - News Channel Nebraska
- Global Potassium Chloride Industry (2020 to 2027) - Market Trajectory & Analytics - Business Wire
- Potassium Chloride Market - Global Analysis and Forecast - Maximize Market Research
- Potassium Chloride Market Size, Growth, Trend & Share | 2033 - Future Market Insights
- Annual Report 2020 - K+S Aktiengesellschaft - K+S Aktiengesellschaft