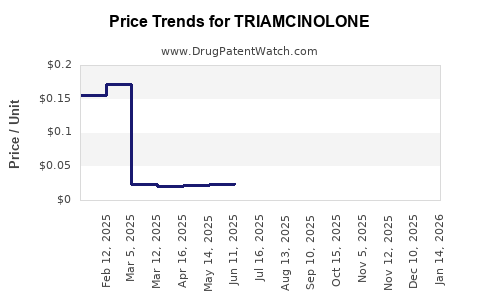
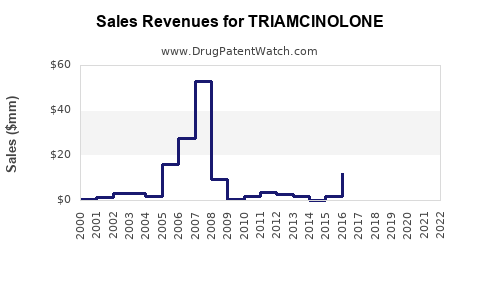
Introduction to Triamcinolone
Triamcinolone, a potent corticosteroid, is widely used in the treatment of various chronic inflammatory diseases, including asthma, arthritis, allergies, and several dermatological conditions. Its anti-inflammatory and immunosuppressive properties make it a crucial component in modern healthcare.
Market Size and Growth Projections
The global triamcinolone market was valued at $0.9 billion in 2023 and is projected to reach $1.3 billion by 2033, growing at a Compound Annual Growth Rate (CAGR) of 3.8% from 2024 to 2033[1].
Key Drivers of Market Growth
Increasing Prevalence of Chronic Diseases
The rise in chronic inflammatory diseases such as asthma, allergies, rheumatoid arthritis, and various dermatological disorders is a significant driver of the triamcinolone market. These conditions are becoming more common due to factors like aging populations, unhealthy lifestyles, and environmental changes[1].
Growing Geriatric Population
The global population is aging, leading to an increase in age-related chronic and inflammatory diseases. Elderly individuals are more prone to these ailments due to the natural decline in immune function and the cumulative wear and tear on the body’s systems over time. For instance, the prevalence of allergic diseases in the elderly is estimated to be around 5-10%[1].
Expansion of Healthcare Infrastructure
The growth in healthcare infrastructure, particularly in developing regions, is enhancing access to triamcinolone and other healthcare services. This expansion is fueled by rising healthcare expenditure and improving medical infrastructure[1].
Market Segmentation
By Type
The triamcinolone market is segmented into various forms, including creams, injections, and inhalers. Each form caters to different applications and patient needs, contributing to the market's diversity and growth[1].
By Application
The market is divided into several applications, such as eczema, dermatitis, allergies, psoriasis, and others. The versatility of triamcinolone in treating a wide range of conditions is a key factor in its market success[1].
By Distribution Channel
Triamcinolone is distributed through drug stores, hospital pharmacies, and online pharmacies. The convenience and accessibility offered by these channels contribute to the market's growth[1].
Regional Market Outlook
North America
North America dominated the triamcinolone market share in 2023, driven by the high prevalence of chronic conditions, advanced healthcare systems, and significant investment in research and development. The U.S. is a major contributor to this dominance due to its large patient population and widespread access to healthcare services[1].
Asia-Pacific
The Asia-Pacific region is experiencing rapid growth, driven by rising healthcare expenditure, improving medical infrastructure, and increasing awareness about chronic diseases and their treatments. Countries like China, Japan, and India are at the forefront of this growth, driven by large populations and developing healthcare infrastructure[1].
Europe and LAMEA
Developed regions like Europe and emerging economies in LAMEA also show significant growth prospects. Europe boasts well-established dermatology markets and adheres to stringent environmental regulations, while LAMEA regions are expanding their accessibility to dermatological treatments[4].
Key Market Players
Major players in the triamcinolone market include Novartis AG, Mylan N.V., Lupin, Teva Pharmaceuticals, Akorn Inc, Cosette Pharmaceuticals, Inc, Bristol Myers Squibb Company, Glenmark Pharmaceuticals, Taro Pharmaceutical Industries, and Sun Pharmaceutical Industries Limited. These companies are driving innovation and market expansion through their extensive product portfolios and strategic acquisitions[1].
Impact of Acquisitions and Mergers
The acquisition of Flexion Therapeutics by Pacira BioSciences in November 2021 added ZILRETTA (triamcinolone acetonide extended-release injectable suspension) to Pacira's commercial offerings. This move has enhanced the company's presence in the non-opioid pain management and regenerative health solutions market[2].
Financial Performance and Challenges
Revenue and Sales
Pacira BioSciences reported preliminary net product sales of $50.2 million for July 2022, which includes sales of ZILRETTA. The company's revenues have been impacted by COVID-19 and related challenges, including the postponement or suspension of elective surgical procedures. However, the introduction of vaccines and the lessening of elective surgery restrictions have helped in the recovery[2].
COVID-19 Impact
The COVID-19 pandemic has had a significant impact on the elective surgery market, affecting the sales of triamcinolone and other related products. Despite the challenges, companies are reporting monthly intra-quarter unaudited net product sales to provide greater transparency and are working to advance their product pipelines[2].
Market Trends and Opportunities
Increasing Awareness and Healthcare Expenditure
The rising awareness about the beneficial outcomes of using topical corticosteroids like triamcinolone for managing skin issues is driving market growth. Additionally, increasing healthcare expenditure, especially in emerging economies, is expanding the accessibility to dermatological treatments[4].
Technological Advancements
The development of extended-release formulations, such as ZILRETTA, is a significant trend in the triamcinolone market. These advancements improve patient compliance and treatment efficacy, contributing to market growth[2].
Restraints and Challenges
Regulatory Policies
Variations in regulatory policies across different regions can affect the demand for triamcinolone. Developed regions adhere to stringent environmental regulations, which can impact the market dynamics[4].
COVID-19 Uncertainty
The ongoing impact of COVID-19 on elective surgeries and global economic conditions remains a challenge for the market. Companies are navigating these uncertainties by providing regular sales updates and focusing on pipeline development[2].
Consumer Behavior and ESG Analysis
Consumer Awareness
Increasing consumer awareness about chronic diseases and their treatments is driving the demand for triamcinolone. Patients are seeking effective and convenient treatment options, which is reflected in the market's growth[4].
Environmental Considerations
The use of chlorofluorocarbons in some triamcinolone formulations is subject to environmental regulations. Companies are adapting to these regulations, which can influence market dynamics and product development[4].
Key Takeaways
- The global triamcinolone market is projected to grow from $0.9 billion in 2023 to $1.3 billion by 2033, with a CAGR of 3.8%.
- The increasing prevalence of chronic inflammatory diseases and the growing geriatric population are key drivers of market growth.
- North America dominates the market, but the Asia-Pacific region is experiencing rapid growth.
- Major players are driving innovation through acquisitions and product development.
- The COVID-19 pandemic has impacted the market, but companies are adapting and focusing on pipeline development.
FAQs
What is the projected market size of triamcinolone by 2033?
The global triamcinolone market is projected to reach $1.3 billion by 2033[1].
What are the key drivers of the triamcinolone market?
The key drivers include the increasing prevalence of chronic inflammatory diseases, the growing geriatric population, and the expansion of healthcare infrastructure[1].
Which region dominates the triamcinolone market?
North America dominates the triamcinolone market, primarily due to the high prevalence of chronic conditions and advanced healthcare systems[1].
What are the different forms of triamcinolone available in the market?
Triamcinolone is available in various forms, including creams, injections, and inhalers[1].
How has the COVID-19 pandemic impacted the triamcinolone market?
The COVID-19 pandemic has impacted the elective surgery market, affecting the sales of triamcinolone and related products. However, companies are adapting and focusing on pipeline development to mitigate these effects[2].
Sources
- Allied Market Research: Triamcinolone Market Statistics, Trends | Forecast - 2033
- Pacira BioSciences: Pacira BioSciences Reports Preliminary Net Product Sales of $50.2 Million for July 2022
- Cognitive Market Research: Global Triamcinolone Acetonide Ointment Market Report 2024
- Grand View Research: Triamcinolone Ointment Chlorofluorocarbons Market Report, 2030