Introduction
VYTORIN, a combination drug of ezetimibe and simvastatin, is a significant player in the market for treating high cholesterol and reducing cardiovascular risk. Here, we will delve into the market dynamics and financial trajectory of VYTORIN, highlighting its effectiveness, market performance, and financial impact.
Effectiveness of VYTORIN
VYTORIN has been proven effective in several clinical studies for treating high cholesterol and reducing cardiovascular events.
Clinical Studies
In a 12-week study, VYTORIN demonstrated superior efficacy compared to its individual components, simvastatin and ezetimibe, and a placebo. The study showed that VYTORIN reduced total cholesterol by 38%, LDL cholesterol by 53%, triglycerides by 24%, and increased HDL cholesterol by 7%[1].
The IMPROVE-IT study, involving over 18,000 patients with acute coronary syndromes, further reinforced VYTORIN's benefits. It showed that VYTORIN reduced total cardiovascular events by 9% compared to simvastatin alone, including a 6.4% reduction in initial events and a 12% reduction in recurrent events[4].
Market Performance
VYTORIN's market performance is closely tied to its efficacy and the demand for cholesterol-lowering medications.
Sales and Revenue
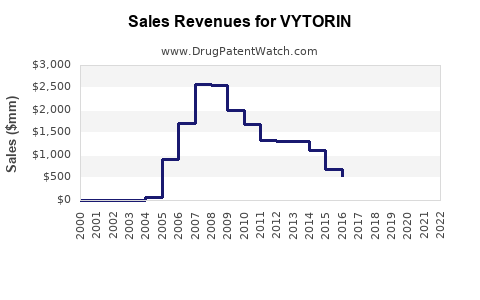
VYTORIN is part of Merck's (known as MSD outside the U.S. and Canada) pharmaceutical portfolio. While specific sales figures for VYTORIN are not always detailed separately, it contributes to the overall revenue of Merck's cardiovascular division.
In 2007, Merck's cardiovascular products, including VYTORIN, played a significant role in offsetting revenue declines from other products. The company's pharmaceutical sales, which include VYTORIN, were $19.6 billion in 2007[3].
Competitive Landscape
VYTORIN competes in a crowded market with other statins and cholesterol-lowering drugs. However, its combination of ezetimibe and simvastatin offers a unique advantage, as seen in clinical studies. The drug's performance in reducing both initial and recurrent cardiovascular events positions it favorably against competitors like rosuvastatin (Crestor)[1][4].
Financial Trajectory
Merck's financial performance provides insight into the economic impact of VYTORIN.
Annual Sales and Revenue
Merck's overall pharmaceutical sales have shown steady growth. In 2018, Merck's worldwide sales were $42.3 billion, a 5% increase from 2017. While VYTORIN's specific sales are not detailed, the growth in Merck's pharmaceutical segment indicates a positive financial trajectory for the drug[2].
Profitability and Shareholder Returns
Merck's profitability and shareholder returns are also indicative of the financial health of its products, including VYTORIN. In 2018, Merck returned $14 billion to shareholders through share repurchases and dividends, reflecting the company's strong financial position[2].
Research and Development
Merck continues to invest in research and development, which includes further studies and analyses on VYTORIN. The IMPROVE-IT study, for example, was a significant investment that provided valuable data on VYTORIN's efficacy in reducing cardiovascular events[4].
Regulatory and Safety Considerations
VYTORIN's market dynamics are also influenced by regulatory and safety considerations.
FDA Approval and Safety Profile
VYTORIN is FDA-approved for treating high cholesterol in adults and children aged 10 and older. However, it comes with certain safety warnings, such as the risk of myopathy and rhabdomyolysis, especially when combined with strong CYP3A4 inhibitors[1][4].
Market Trends and Future Outlook
The market for cholesterol-lowering medications is driven by increasing awareness of cardiovascular health and the need for effective treatments.
Growing Demand for Cardiovascular Treatments
The demand for drugs like VYTORIN is expected to grow as the global population ages and the prevalence of cardiovascular diseases increases. Clinical studies like IMPROVE-IT continue to support the use of VYTORIN in high-risk patients, which could further boost its market presence[4].
Competitive Innovations
The pharmaceutical industry is highly competitive, with continuous innovation in cardiovascular treatments. VYTORIN's unique combination and proven efficacy will need to be maintained or improved to stay competitive in the market.
Key Takeaways
- Clinical Efficacy: VYTORIN has been shown to be highly effective in reducing cholesterol levels and cardiovascular events.
- Market Performance: The drug contributes significantly to Merck's pharmaceutical revenue, although specific figures are not always detailed.
- Financial Trajectory: Merck's overall financial performance indicates a positive trajectory for VYTORIN, with strong sales and profitability.
- Regulatory and Safety: VYTORIN is FDA-approved but comes with specific safety warnings.
- Market Trends: The demand for effective cholesterol-lowering medications is expected to grow, supporting VYTORIN's market presence.
FAQs
What is VYTORIN used for?
VYTORIN is used to treat high cholesterol caused by primary hyperlipidemia in adults and children aged 10 and older.
How effective is VYTORIN in reducing cholesterol?
VYTORIN reduces total cholesterol by an average of 38%, LDL cholesterol by 53%, triglycerides by 24%, and increases HDL cholesterol by 7% in clinical studies[1].
What are the key findings of the IMPROVE-IT study?
The IMPROVE-IT study showed that VYTORIN reduced total cardiovascular events by 9% compared to simvastatin alone, including a 6.4% reduction in initial events and a 12% reduction in recurrent events[4].
What are the potential side effects of VYTORIN?
VYTORIN can cause myopathy, rhabdomyolysis, and other muscle-related issues, especially when combined with strong CYP3A4 inhibitors[1][4].
How does VYTORIN compare to other cholesterol-lowering medications?
VYTORIN's combination of ezetimibe and simvastatin offers unique benefits, particularly in reducing both initial and recurrent cardiovascular events, positioning it favorably against competitors like rosuvastatin (Crestor)[1][4].
Sources
- Medical News Today: Vytorin: Side effects, uses, dosage, interactions, and more.
- Merck: Merck Announces Fourth-Quarter and Full-Year 2018 Financial Results.
- Annual Reports: Merck & Co., Inc. - Annual Reports.
- Merck: New Analysis from Investigational IMPROVE-IT Study Shows VYTORIN® (ezetimibe/simvastatin) Reduced Total (Initial and Recurrent) Cardiovascular Events More than Simvastatin Alone in Patients Presenting with Acute Coronary Syndromes.